January 5, 2021 at 10:08 pm | Updated January 5, 2021 at 10:08 pm | 6 min read
Scientists are constantly searching for reliable and affordable sources of medicines. It is well known that many plants have medicinal value and trace amounts of the phytochemicals of interest. However, the production of medicines from these plants requires that harvesting and extraction be in amounts that allow the process to remain sustainable. One study that focused on improving phenolic content from the common opuntia used a multiple gas analyzer to make the research easier.
Increasing Biosynthesis of Phenolic Compounds
Opuntia ficus-indica (L.) Mill., or the prickly pear, belongs to the family Cactaceae and is common in the tropics and sub-tropics. In Mexico, opuntia is consumed as a fruit and also as a treatment for ulcers, high levels of cholesterol, and even cancer.
Its medicinal value comes from phytochemicals, such as flavonoids—quercetin, kaempferol, and isorhamnetin—and ascorbic acid. These compounds are secondary metabolites found in the pulp and skin of opuntia.
Subscribe to receive our monthly round-up of articles.
Scientists Ortega-Hernández, Nair, Welti-Chanes, Cisneros-Zevallos, and Jacobo-Velázquez wanted to see if a combination of wounding and ultraviolet B (UVB) radiation could increase the accumulation of these phytochemicals and find out the role of a few key enzymes that could be aiding the process.
Prickly pear fruits were kept in ultraviolet-B light chambers made of steel and were treated with 6.4 W·m−2 UVB for 0 and 15 minutes. Each treatment had 20 whole and 12 wounded opuntia fruits.
After the experiment, the level of ascorbic acid and flavonoids in the fruits was determined immediately and stored for 24 hours. Enzyme activity of phenylalanine ammonia-lyase (PAL) and L-galactono-γ-lactone dehydrogenase (GalLDH) was determined 0 and 12 hours after storage.
In another part of the experiment, the scientists wanted to determine the respiration rate by measuring carbon dioxide (CO2) and the production of ethylene-related volatile organic compounds (VOCs) for three whole and five wounded samples, 0, 6, 12, 18, and 24 hours after storage at 16°C. Hence, they needed to measure ethylene and carbon dioxide levels non-destructively, as readings had to be repeated five times using the same samples.
Challenge: Combining Analysis of Two Gases
There are several methods to measure respiration and ethylene non-destructively; however only a few will measure both gases with the same efficiency.
Respiration of a fruit during the experiment can be measured by closed systems and flow systems. In the closed system, samples are kept in a sealed container, and concentrations of carbon dioxide and oxygen (O2) are measured before and after a given time, usually an hour. Though the setup is easy, the accumulation of ethylene and CO2 and depletion of O2 will affect the rate of respiration. There have to be enough gas differences so that it can be recorded, but not too high to affect the experiment. Therefore, this method is not suitable for high respiring fruits.
In the flow system, the sample is kept in a closed container with an inlet and outlet, and the gas at both ends is periodically measured. Estimation is far more difficult in this method because air is continuously flowing and is not suitable for low respiring fruits, as low levels are difficult to determine.
In both cases, CO2 is estimated by gas chromatography and infrared spectroscopy.
Ethylene can also be estimated by miniature gas chromatography; however, the instruments have limited sensitivity and repeatability as the method is sensitive to changes in environmental conditions.
Infrared spectroscopy is more suitable to measure CO2 than ethylene.
The scientists needed a method that was not difficult or time consuming to set up and operate. Using two different methods for the 40 samplings required in the experiment would add a considerable amount of work, so an instrument that could measure both gases was preferred.
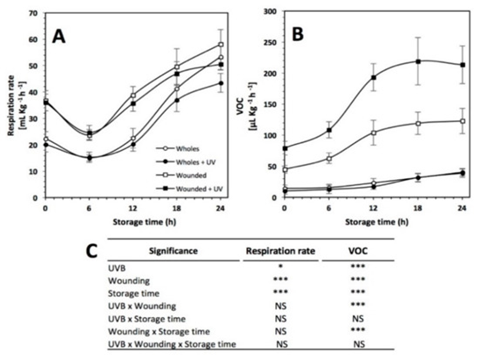
Figure 1.: “(A) Respiration rate and (B) volatile organic compounds (VOCs) production in untreated red prickly pear, treated with UVB light (6.4 W·m−2) for 15 min alone, treated with wounding stress alone, and treated with both UVB light and wounding stress during storage time (0, 6, 12, 18, and 24 h) at 16°C. (C) Full factorial analyses of variance showing the main effects and interactions of the variables evaluated,” Ortega-Hernández et al. 2019. (Image credits: Int. J. Mol. Sci. 2019, 20(21), 5327; https://doi.org/10.3390/ijms20215327)
Solution: The F-950 Three Gas Analyzer
Finding an instrument that combines various estimation methods for gases is not easy. Most instruments measure only one gas. However, the scientists found this combination in the F-950 Three Gas Analyzer, produced by Felix Instruments Applied Food Science.
The F-950 Three Gas Analyzer measures a wide range of both ethylene and CO2, in addition to O2. It is a small portable tool that is also very precise.
Ortega-Hernández and her colleagues first kept the samples in separate plastic jars for an hour at room temperature (22°C) to allow the gases to accumulate. They collected 15 ml of gas from the containers with the included needle probe connected to the intake of the F-950.
As the instrument is made to measure ethylene in the food supply chain, it comes with a PolarCept!™ hydro filter that removes hydrocarbons other than ethylene, so the scientists could be sure the experiment was not compromised by other contaminant gases.
The scientists were able to get readings of the levels of both gases within a minute, simultaneously.
A Single Gas Analyzer Gave Precise and Easy Measurements
If the scientists had used two instruments or other conventional laboratory setups, they would have been unable to make simultaneous measurements. Moreover, the gas reading would have been influenced by changes in temperatures and humidity, which can affect the rate of both respiration and ethylene production.
The F-950 uses the Electrochemical method, which is the most efficient method of estimating ethylene and can measure a wide range from 0-200 ppm.
Carbon dioxide is measured by two methods: the infrared sensor and pyroelectric detector. The F-950 measures CO2 from 0-100%.
The F-950 can be used in a wide range of temperatures and humidity, and it weighs less than one kilo. It has a battery that lasts for eight hours, so experiments can be completed on the same day or within an hour or two.
The readings are recorded and stored in the 16 GB SD card that can be transferred later to the computer for direct statistical analysis, sparing time needed for data entry.
Wounding and UVB Can Increase Biosynthesis
The scientists were able to show that the combination of UVB radiation and wounding induced synthesis and accumulation of the desired metabolites.
Respiration rate increased due to wounding, and production of ethylene increased due to the dual stress of wounding and UVB radiation, as shown in Figure 1.
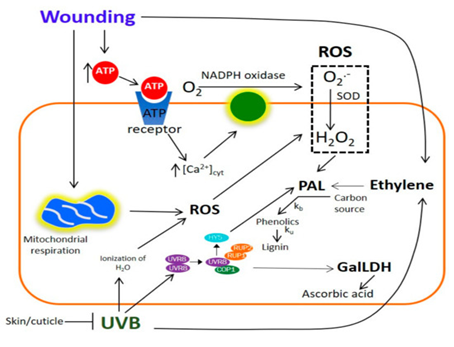
Figure 2. “Proposed model explaining the combined effect of wounding and UVB radiation stresses on the accumulation of phenolic compounds and ascorbic acid in prickly pear. Abbreviations: L-galactono-γ-lactone dehydrogenase (GalLDH); UV RESISTANCE LOCUS 8 photoreceptor (UVR8); CONSTITUTIVELY PHOTOMORPHOGENIC 1 (COP1); REPRESSOR OF UV-B PHOTOMORPHOGENESIS 1 and 2 (RUP1/RUP2); transcription factor ELONGATED HYPOCOTYL5 (HY5); reactive oxygen species (ROS); phenylalanine ammonia-lyase (PAL),” Ortega-Hernández et al. 2019. (Image credits: Int. J. Mol. Sci. 2019, 20(21), 5327; https://doi.org/10.3390/ijms20215327)
Treating whole fruits with UVB for 15 minutes produced the phenolic compounds in the pulp and skin. Quercetin, rosmarinic acid, sinapic acid, and sinapoyl malate increased by 709.8%, 442.8%, 570.2%, 439.9%, and 186.2%, respectively, compared to pre-treatment levels. The phenols were produced in response to phenylalanine ammonia-lyase (PAL) activity, which increased due to radiation in both wounded and whole fruits; see Figure 2.
Ascorbic acid was produced when the peel was removed and the inner tissues were exposed to UVB radiation. This was a result of the activity of the enzyme l-galactono-γ-lactone dehydrogenase (GalLDH), which increased when exposed pulp in wounded opuntia were treated with UVB; see Figure 2.
The scientists concluded that the ethylene and ROS produced due to stress acted as signalling molecules, resulting in the production of phenolic compounds. Hence, stressed opuntia can be used to extract the desired phytochemicals for pharmaceutical and dietary supplementary use.
Making Technology Accessible
The combination of several methods of estimation for three gases in one tool, such as the F-950, is a vital asset. These tools have improved research efforts not only in crop science, but also in potentially expanding the use of naturally sourced medicines.
—
Vijayalaxmi Kinhal
Science Writer, CID Bio-Science
Ph.D. Ecology and Environmental Science, B.Sc Agriculture
Feature image courtesy of Pikist.
Sources
Fonseca, S. C., Oliveira, F. A., & Brecht, J. K. (2002). Modelling respiration rate of fresh fruits and vegetables for modified atmosphere packages: A review. Journal of Food Engineering, 52(2), 99-119. doi:10.1016/s0260-8774(01)00106-6
Kader, A.A., & Saltveit, M.E. (n.d.). Respiration and Gas Exchange. Retrieved from https://irrec.ifas.ufl.edu/postharvest/HOS_5085C/Reading%20Assignments/BartzBrecht-2-Respiration%20and%20Gas%20Exchange.pdf
Ortega-Hernández, E., Vimal Nair, V., Jorge Welti-Chanes, J., Luis Cisneros-Zevallos, L., & Jacobo-Velázquez, D.A. (2019). Wounding and UVB Light Synergistically Induce the Biosynthesis of Phenolic Compounds and Ascorbic Acid in Red Prickly Pears (Opuntia ficus-indica cv. Rojo Vigor). Int. J. Mol. Sci., 20(21), 5327; https://doi.org/10.3390/ijms20215327
Related Products
- F-751 Grape Quality Meter
- Custom Model Building
- F-910 AccuStore
- F-751 Melon Quality Meter
- F-751 Kiwifruit Quality Meter
- F-750 Produce Quality Meter
- F-751 Avocado Quality Meter
- F-751 Mango Quality Meter
- F-900 Portable Ethylene Analyzer
- F-950 Three Gas Analyzer
- F-920 Check It! Gas Analyzer
- F-960 Ripen It! Gas Analyzer
- F-940 Store It! Gas Analyzer
Most Popular Articles
- Spectrophotometry in 2023
- NIR Applications in Agriculture – Everything…
- The Importance of Food Quality Testing
- The 5 Most Important Parameters in Produce Quality Control
- Melon Fruit: Quality, Production & Physiology
- Fruit Respiration Impact on Fruit Quality
- Guide to Fresh Fruit Quality Control
- Liquid Spectrophotometry & Food Industry Applications
- Ethylene (C2H4) – Ripening, Crops & Agriculture
- Active Packaging: What it is and why it’s important






