November 3, 2020 at 12:19 am | Updated November 3, 2020 at 12:19 am | 6 min read
Ethylene (C2H4), a hormone produced in the roots and shoots of a plant, is responsible for the ripening processes in climacteric fruits, germination of seeds, elongation of the stem, and senescence of leaves. Depending on the species, ethylene can also encourage or inhibit flowering, pollen and ovule development, flower opening, and senescence. Moreover, ethylene can also be produced in increased quantities in response to stress. The F-900 Portable Ethylene Analyzer from Felix Instruments Applied Science is one of the few instruments useful for on-site experiments to examine ethylene effects on plant dynamics
Ethylene Impact on Coffee
Flowering is important not only for plants but also for people as it is a precursor to much of the food that we consume. Coffee is an important cash crop and is considered to be the second most valuable traded agricultural commodity.
A short period of water deficit followed by rainfall is one of the environmental features necessary to trigger the opening and development of flower buds. Thus, in places like Brazil, with a well-defined dry season, anthesis occurs only two to four times; whereas, in equatorial regions, like Ethiopia, Colombia, etc., coffee has many episodes of anthesis and coffee fruit development. Due to the multiple “rounds” of harvesting in such equatorial regions, developing a greater understanding of control mechanisms in the flowering process is crucial for production consistency and profitability.
Subscribe to receive our monthly round-up of articles.
The formation of flower buds is not synchronous or simultaneous. Flower buds start and then complete differentiation and development, after which growth stops and the buds enter a dormant state. Usually, this coincides with the beginning of the dry season in Brazil, so more buds have the time to complete their development. By the time the rain arrives, there are more flowers ready to open.
Many internal physiological factors in plants are also important for anthesis, and scientists speculate that ethylene could be one of them.
Ethylene gas diffuses through intercellular spaces and moves within the tissues. Several receptors form the ethylene signaling pathway, and their concentrations determine how sensitive tissue is to ethylene. Drought can affect the expression of genes that regulate the formation of these receptors.
Therefore, there are various ways in which drought could be impacting flowering in coffee.
A team of scientists, Lima et al., decided to check how ethylene was affecting coffee anthesis in Brazil. To regulate ethylene production in their experiment, they used 1-Methylcyclopropene (1-MCP), which is a growth regulator known to inhibit ethylene action. There were two parts to the experiment: one in the greenhouse and the other in the field.
Greenhouse Study
The first part of the study tested the response of coffee plants to water dynamics. The scientists chose 6-month old coffee seedlings, since the species flowers only after the third year. They were divided into three treatments: well-watered plants, water-deficit plants, and plants rewatered after water deficit.
The soil water potential (Ψpd) was measured before data collection, as plucking leaves increases ethylene production.
Gas exchanges of carbon assimilation rate and stomatal conductance were recorded using a portable infrared gas analyzer, along with the leaf water potential (Ψpd). Plants were sampled after 0, 2, 4, 6, 12, 24, and 48 hours of re-watering. The leaves were then cleaned and frozen in liquid nitrogen for gene expression experiments.
0, 2, 6, and 24 hours after water treatments were imposed, Reverse Transcription – Quantitative Polymerase Chain Reaction was used to study the expression of genes involved in ethylene synthesis and signalling. Genes controlling one ACC synthase (CaACS1-like), two ACC oxidases (CaACO1-like and CaACO4-like), and one ethylene receptor (CaETR4-like) from shoots and roots were studied.
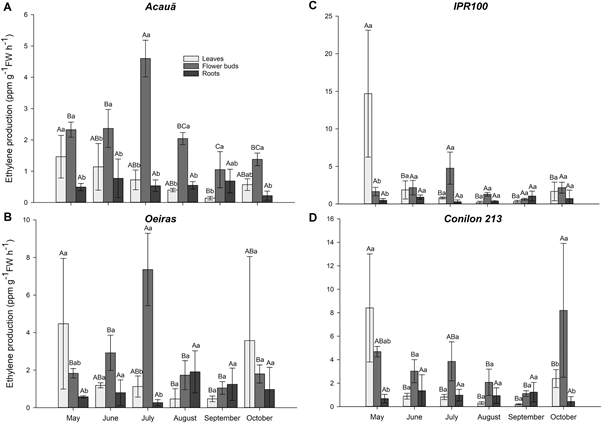
Figure 1: “Ethylene production from leaves, flower buds and roots of the Acauã (A), Oeiras (B), IPR100 (C), Conilon 213 (D), and coffee cultivars analyzed in Field experiment I, from May (before the dry season) to October (rainy season),” Lima et al. 2021. (Image credits: https://doi.org/10.1016/j.envexpbot.2020.104289)
Field Experiments
There were three field experiments:
1. The first monitored ethylene production in four cultivars of coffee: three from Coffea arabica (Acau˜a, IPR100, and Oeiras) and one of Coffea canephora (Conilon 213). Each of the cultivars was a treatment, and the root, leaves, and bud tissue were tested for ethylene from May to October. The last sampling of buds was during flowering in September after rainfall, and the last sampling for leaves and roots ended two weeks later (Figure 1).
Ethylene was measured from the headspace gas using the F-900 Portable Ethylene Analyzer, manufactured by Felix Instruments – Applied Food Science. The tissue material was first incubated in two vials, and 2.5 ml of the gas released was collected by a 10 ml plastic syringe and injected into the F-900. The ethylene was measured in the GC Emulation Mode, and results were obtained in real-time within a few minutes. The plant material in the vials was weighed and the gas released was expressed as ppm g-1h-1.
Plant water stress was measured prior to leaf water potential, just as in the greenhouse experiments.
2. The second field experiment was about the effect of ethylene on flowering. Eight-year-old coffee trees from a second plantation were tested in August just before rainfall.
There were seven treatments in all: five based on the level of Harvista 1-MCP gas concentrations, the sixth set sprayed with an organosilicon surfactant (BreakThru), and the last was the control that was not sprayed. In the end, the flower buds that opened or progressed to the anthesis stage were counted.
3. Physiological and molecular changes due to 1- MCP were measured in the third experiment, conducted in the second plantation, 2, 24, and 48 hours after 1-MCP application. Carbon assimilation (photosynthesis) and stomatal conductance, as well as gene expression of the ethylene synthesis and signaling genes, were measured.

Figure 2: “Representation of the anthesis induction from Field experiment II in response to Harvista application. Control plants (A) did not show anthesis induction, as with the BTH, MCP1, MCP2, and MCP3 treatments (not shown), while plants from the MCP4 and MCP5 (B) treatments showed a significant increase in flower bud size (B), with flower opening taking place 12 days after Harvista application (C),” Lima et al. 2021. (Image credits: https://doi.org/10.1016/j.envexpbot.2020.104289)
The two treatments, soil drying followed by rewatering and chemical treatment with 1-MCP, enhanced anthesis in coffee by influencing ethylene production and its reception pathways.
As expected, leaf and flower bud ethylene production decreased during the dry season. Due to the down-regulation of CaACO1-like gene for ethylene signaling, the ethylene content in shoots decreased. Root ethylene remained unaffected by water status.
Rewatering increased shoot ethylene production due to the up-regulation of CaACO1-like gene and possibly due to transport of root ethylene to the shoots.
Ethylene is also important in regulating the stomatal opening, which is smaller in rewatered plants compared to well-watered plants.
The field experiments showed that ethylene increase per se is not enough to lead to coffee anthesis. Ethylene levels do increase in the shoot due to rewatering and 1-MCP due to the up-regulation of the genes for its production; see Figure 2. However, a change in sensitivity or signaling pathways is also important. When ethylene levels fall, the ethylene sensitivity increases due to the down-regulation of the ethylene receptor, so less ethylene is fixed and more is available for the anthesis.
Novel Finds
This study, published in 2020, was the first in the world that connected changes in ethylene production and the expression of genes to the flowering that occurs at rainfall after a drought in any woody species. It was also first in showing that 1-MCP can be used to trigger anthesis, in the absence of rainfall, after a drought. The many breakthroughs achieved in this one study were possible due to the availability of portable tools, like the F-900, that can be taken to the field. In addition, the electrochemical sensor technology in the F-900 is faster, more sensitive, and cheaper than gas-chromatography, one of the older alternatives.
—
Vijayalaxmi Kinhal
Science Writer, CID Bio-Science
Ph.D. Ecology and Environmental Science, B.Sc Agriculture
Feature image courtesy of Niek van Son
Sources
Lima, A., Santos, I., Lopez, M., et al. (2021). Drought and re-watering modify ethylene production and sensitivity, and are associated with coffee anthesis. Authorea, 181. DOI: 10.22541/au.158981705.57565156
Cristescu, S.M., Mandon, J., Arslanov, D., et al. (2013). Current methods for detecting ethylene in plants. Ann Bot, 111: 347-360. DOI: 10.1093/aob/mcs259.
Related Products
- F-751 Grape Quality Meter
- Custom Model Building
- F-910 AccuStore
- F-751 Melon Quality Meter
- F-751 Kiwifruit Quality Meter
- F-750 Produce Quality Meter
- F-751 Avocado Quality Meter
- F-751 Mango Quality Meter
- F-900 Portable Ethylene Analyzer
- F-950 Three Gas Analyzer
- F-920 Check It! Gas Analyzer
- F-960 Ripen It! Gas Analyzer
- F-940 Store It! Gas Analyzer
Most Popular Articles
- Spectrophotometry in 2023
- NIR Applications in Agriculture – Everything…
- The Importance of Food Quality Testing
- The 5 Most Important Parameters in Produce Quality Control
- Melon Fruit: Quality, Production & Physiology
- Fruit Respiration Impact on Fruit Quality
- Guide to Fresh Fruit Quality Control
- Liquid Spectrophotometry & Food Industry Applications
- Ethylene (C2H4) – Ripening, Crops & Agriculture
- Active Packaging: What it is and why it’s important






